
WOLLASTONITE |
Pyroxenoid Group of Minerals |
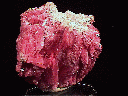
RHODONITE |

RHODONITE |

LARIMAR |

PECTOLITE |
The pyroxenoid minerals are inosilicates of the general formula XSiO3. The X, represents ions such as calcium, sodium, manganese, iron and magnesium. The pyroxenoid's structure is similar to the Pyroxene Group's structure. However, the chains in the pyroxenoid structures are more . . . "kinked"! The typical pyroxene structure contains chains of SiO3 tetrahedrons that every other one alternates from the left side to the right side of the chain. Each of the tetrahedrons has one flat edge on the "base" of the structure as if the chain were a chain of connected three sided pyramids on a flat desert. The orderliness of the tetrahedrons means that they repeat every three tetrahedrons, ie. left-right-left.
In pyroxenoids, the chain is more erratic, twisted, almost helical in nature. The repeat interval is variable and depends on the particular pyroxenoid but is always longer than a pyroxene. It is this erratic nature of the pyroxenoid chains that is primarily responsible for the very low symmetry, usually triclinic; bar 1.
These are the more common minerals of the Pyroxenoid Group:
Bustamite (Calcium Manganese Silicate)Parawollastonite (Calcium Silicate)- Pectolite (Sodium Calcium Silicate Hydroxide)
- Rhodonite (Manganese Iron Magnesium Calcium Silicate)
- Wollastonite (Calcium Silicate)











