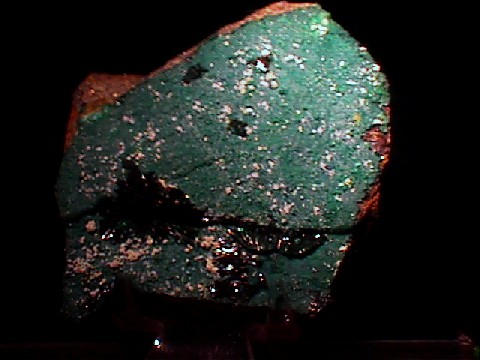- Chemistry: Cu2Cl(OH)3, Copper Chloride Hydroxide.
- Class: Halides
- Subclass: Oxy-halides
- Uses: as a minor ore of copper and as mineral specimens.
Specimens
Atacamite forms in arid climates where copper minerals are exposed to oxidation. The Atacama Desert, from where atacamite gets its name, is one of the driest places in the world. Atacamite has a deep green color and is often associated with many other rare and colorful minerals such as chyrsocolla, brochantite, pseudomalachite, connellite, linarite, caledonite, libethenite, cornetite, cuprite and malachite. Some of these specimens can be quite beautiful, with a combination of blue, green, and red hues. Atacamite is a wonderful and interesting addition to someones collect.
PHYSICAL CHARACTERISTICS:
- Color is dark green.
- Luster is vitreous.
- Transparency: Crystals are transparent to translucent.
- Crystal System is Orthorhombic; 2/m2/m2/m
- Crystal Habits include slender striated crystals that can be accicular to fiberous.
- Cleavage is perfect in one direction.
- Fracture is splintery.
- Hardness is 3 - 3.5
- Specific Gravity is approximately 3.75+ (above average)
- Streak is pale green.
- Associated Minerals include limonite, chyrsocolla, brochantite, pseudomalachite, connellite, linarite, caledonite, libethenite, cornetite, cuprite and malachite.
- Other Characteristics: crystals are vertically striated.
- Notable Occurrences include Atacama Desert, Chile; Mt. Vesuvius, Italy; Wallaroo, Australia; Mexico and Pinal County, Arizona, USA.
- Best Field Indicators are crystal habit, color, associations and localities.