
|
The Gold Group |

|

|
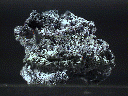

|
|
The Gold Group is composed of metallic elements that have the same crystal structure as gold. The group is composed of only six minerals: gold, silver, copper, mercury, aluminum and lead. People familiar with the periodic table would understand the similarities between gold, copper and silver since as they fall in the same column and thus they share some similar chemical properties. Lead and Aluminum however, don't lie in the same columns as the others but they do crystallize with the same structure as the rest of the group. Mercury is only grudgingly included in this group because it is found alloyed with other members of this group, however it does not by itself form crystals at normal temperates. Minerals of gold, silver and copper are all found in relatively large concentrations within the Earth's crust, but mercury is much rarer, while lead and aluminum are extremely scarce in their native forms.







