THE MINERAL WILLEMITE
THE MINERAL WILLEMITE
- Chemistry: Zn2SiO4, Zinc Silicate.
- Class: Silicates
- Subclass: Nesosilicates
- Group:
Phenakite - Uses: a minor ore of zinc and as mineral specimens.
Specimens
Dispite being discovered at Franklin first, it was a site in Belgium, where the mineral formed small brown crystals, that gave it its name, willemite. It seems that the minerologists at Franklin described the mineral but never named it. In the meantime the minerologists at Belgium named the mineral after William I of Belgium.
Willemite is one of the few silicate minerals that have a trigonal symmetry. This symmetry is far more common among carbonates than among silicates. Willemite shares the same symmetry with the emerald green silicate dioptase and the closely related silicate phenakite. Although massive willemite is much more common, some crystals do show the rhombohedral terminations atop hexagonal prisms that is characteristic of it symmetry.
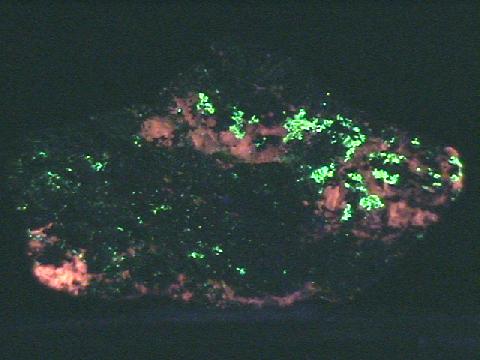
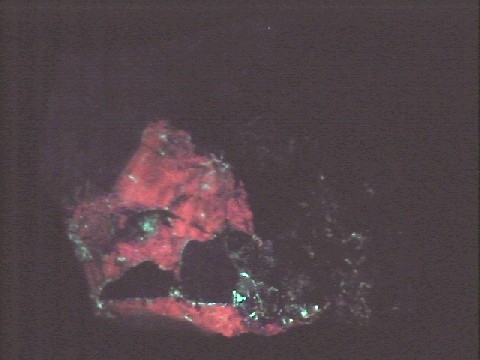
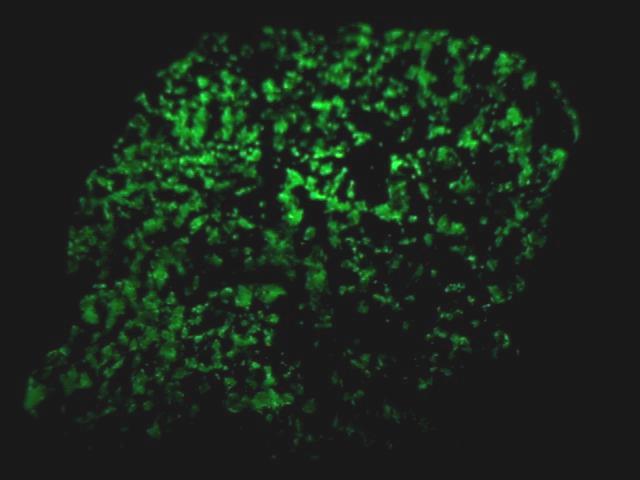
Willemite's great desirability is not so much its use as a zinc ore, its rarity, its associations or even its crystal habits as it is its wonderful fluorescence. Nearly all willemite specimens fluoresce a bright green under ultra-violet light. This fluorescence combined with the red fluorescence of the calcite at Franklin, makes specimens from there even more special. Some willemite specimens will even show phosphorescence. Phosphorescence is the ability of a mineral to glow after the initial light is removed. The mineral has essentially stored the energy of the initial activating light and re-emits light on a delayed basis. Willemite is one of the best examples of a fluorescent mineral and is a must have for any collector interested in this phenomenon.
PHYSICAL CHARACTERISTICS:
- Color is usually colorless or white but can be tinted yellow, blue, red, brown and often green.
- Luster is vitreous to resinous.
- Transparency crystals are transparent to translucent.
- Crystal System: trigonal; bar 3
- Crystal Habits include typically short prismatic crystals although some prismatic crystals can be rather long with sometimes very steep rhombohedral terminations. Also as granular, lamellar and fibrous masses.
- Cleavage in good in one direction (basal)
- Fracture is conchoidal to uneven.
- Hardness is 5.5
- Specific Gravity is approximately 3.9 - 4.2 (above average for non-metallic minerals).
- Streak is white.
- Other Characteristics: strongly fluorescent green and sometimes phosphorescent.
- Associated Minerals are zincite, franklinite, rhodonite, calcite,
greenockite and other rare minerals. - Notable Occurrences include of course Franklin as well as Paterson and Sterling Hill, New Jersey and Mammoth Mine, Tiger, Arizona, USA; Morsnet, Belgium; St. Hilaire, Quebec; Tsumeb, Namibia and Greenland.
- Best Field Indicators are fluorescence, associations, luster, cleavage and crystal habit.