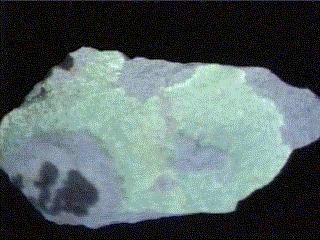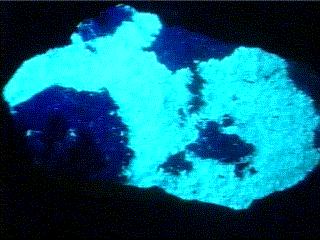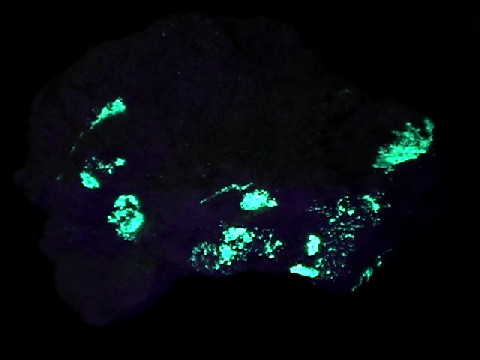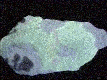
ANDERSONITE
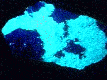
under longwave
UV light
- Chemistry: Na2CaUO2(CO3)3-6H2O, Hydrated Sodium Calcium Uranyl Carbonate
- Class: Carbonates
- Uses: very minor ore of uranium and as mineral specimens.
Specimens
Andersonite's lovely color and unique glow, as well as it's rarity and fluorescence, make it a wonderful mineral for rare mineral collectors. Remember, this is a radioactive mineral and should be stored away from other minerals that are affected by radioactivity and human exposure should definitely be limited.
PHYSICAL CHARACTERISTICS:
- Color is yellow to a yellowish green.
- Luster is vitreous to pearly.
- Transparency crystals are commonly translucent.
- Crystal System is trigonal.
- Crystal Habits include small rhombohedral crystals that have angles close to 90 degrees, making them pseudocubic. Also found commonly as crusts.
- Cleavage is perfect in three directions forming rhombs.
- Fracture is conchoidal.
- Hardness is 2.5
- Specific Gravity is 2.9 (average).
- Streak is pale yellow.
- Associated Minerals are gypsum and other uranium carbonates such as bayleyite, Mg2(UO2)(CO3)3-18H2O and liebigite, Ca2(UO2)(CO3)4-11H2O.
- Other Characteristics: radioactive, and fluoresces bright lemon-yellow under ultraviolet light.
- Notable Occurrences include the Hillside Mine, Bagdad, Arizona and the Jim Thorpe Mine, Pennsylvania, USA.
- Best Field Indicators are its crystal habit, radioactivity, fluorescence and associations.