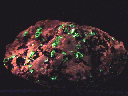
MINERAL LOCALITY:

Franklinite
|
The mines of Franklin and the Sterling Hill Mine at Ogdensburg,
Sussex County in northwestern New Jersey are world famous and deservingly
so. No other site can boast the same assortment of rare and interesting
minerals. Over three hundred different minerals were found at these mines
and most are listed in The
Minerals of Franklin and Sterling Hill Table. Over 60 new minerals
to science were also described from samples taken from these mines, thus
claiming these mines as their type locality (these minerals are shown in
bold in the table). While other great localities can have similarities
with other sites, there simply are no real good parallels with the mineral
assortments of Franklin and Sterling Hill, New Jersey.
The geological reasons for this diversity of minerals is somewhat complex. It involves zinc, manganese and iron rich sediments on a pre-Cambrian sea floor being swept up into a regional orogenic event that created a mountain chain in the approximate position of the current Appalachian Mountains. This event however occurred more than a billion years ago. The origin of the zinc, manganese and iron sediments is theorized to have been manganese nodules and/or sulfide producing "black smokers" that we see along mid-oceanic ridges today. Whatever the case, the manganese and zinc is what drives almost all of the unique and exotic mineral species that are found here. Later contact and regional metamorphism, hydrothermal alterations and weathering produced unusual results and thus a whole "mess" of rare zinc and manganese minerals. The minerals that were found here are unlike those found anywhere else. Unusual manganese and zinc oxides and silicates as well as a few arsenates, are the hallmark of this locality. The primary ore minerals are the franklinite (an iron, zinc and manganese oxide) and willemite (a zinc silicate) and to a lesser extent zincite (a red colored zinc oxide) and hemimorphite (a zinc silicate). Iron is the primary product, in terms of weight, while zinc and manganese are rather significant. When first exploited, it was thought that the ore minerals were magnetite (an iron oxide) and cuprite (a red colored copper oxide), but the ore behaved differently than other magnetite ores in the smelting process and the "cuprite" yielded no copper. Of course most of the "magnetite" turned out to be a new mineral to science, franklinite and the "cuprite" turned out to be zincite, one of the first new minerals identified in the New World and one of many to come from this locality. As the mining continued, more uses and better techniques for the exploitation of zinc and manganese were found and the mine became a boom for the area. The steel, paint and coal industries (needed to smelt the ore) were all positively affected by these mines. The iron/manganese alloys strengthened steel; while the zinc was used in a variety of paints and in certain alloys. Manganese is the typical activator, either as a trace element or as a primary element in the fluorescent mineral's chemistry. The ore body at Franklin and Sterling Hill is surrounded by a marble made up of mostly calcite similar in appearance to the calcite of fluorescent fame. Only a few meters from the ore body however, the calcite is non-fluorescent. It lacks the manganese as an activator. This tells geologist how far the manganese permeated into the surrounding rocks. Several minerals from this site have been cut as gemstones. Many rank as the largest gemstones of their kind in the United States and the world. Although most are not nor have ever been significant on the gemstone markets, they were cut by and for gemstone collectors who seek unusual gemstones. Willemite, zincite, rutile, hodgkinsonite and friedelite are a few of the rare gemstone minerals that have been cut for collectors and museums from here. Sadly, the mines are now closed and filled with water and rock. Only a few mine dumps remain to provide any new material. Two museums are present to educate the public about these remarkable mines and one allows tours into some of the actual mine shafts and both provide mine dump collecting opportunities. It is easy to see that these mines are truly one of the best mineral localities to ever be discovered and specimens from them should be treasured. For a nearly complete list of minerals found at this locality see The Minerals of Franklin and Sterling Hill Table. Much information for this page was provided by Franklin and Sterling Hill website. |